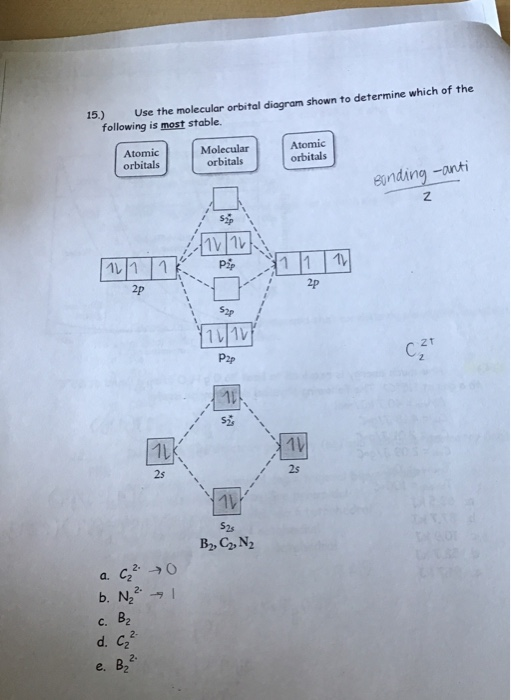
The number of molecular orbitals is the sum of the atomic orbitals contributed by the individual atoms.These orbitals are called anti-bonding orbitals. One of the results of MO theory is that some electrons will be in orbitals that do not lie between the bonding atoms. In the case of some molecules (especially those involving oxygen with one other atom), MO provides an adequate explanation while VSEPR cannot. In most cases, both theories account for the same phenomenon with the same results. The digital watch requires us to do some arithmetic to figure the difference as 15 minutes, which is more precise numerically. The analog watch (with long and short hands) gives us a graphical, qualitative "picture-sense" of the relationship between two times as the "second" or lower-right quarter of a circle. 
We do the same kind of theory application when we use both an analog and digital watch to figure the difference between 12:15pm and 12:30pm. While the VSEPR theory gives us a good general picture of molecules (particularly those with many atoms), the MO theory is used when we want a more detailed, quantified view, especially if we have electrons in excited states. Milliken (the "oil-drop" man to you physicists). The molecular orbital theory is the brain child of Robert S. Sometimes the concept of a pair of electrons shared between only two atoms can't account for all the phenomena we observe, and another theory is necessary. But the close proximity of multiple nuclei in a molecule creates superimposed fields that affect the electrons of the atoms. So far, we've been focussed on two atoms sharing a pair of electrons to form a covalent bond. Optional Website Reading Electrons Among Atoms.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
